Fluorescence Microscopes
Observing Spheroids and Organoids
What are spheroids?
Spheroids are cellular aggregates.
Spheroids can be generated by aggregating and organizing thousands of cells into a sphere using a 3D culture. Spheroids are known for offering higher functional expression levels for longer periods than 2D cell cultures. Because spheroids form a metabolic gradient, they create heterogeneous cellular distribution through interactions between cells and the extracellular matrix (ECM) or between the cells themselves. Spheroids can be generated from various cell types, including neural tissue, embryoid bodies, hepatocytes, tumor tissue, or mammary cells. The structure is physiologically similar to 2D cell cultures, enabling high-degree reproduction of various diseased tissue conditions.
These properties make spheroids a popular choice for screening when evaluating drug efficacy and toxicity. Research into transplanting spheroids into patients as a treatment is also progressing, and the usefulness of spheroids in the treatment of ischemic diseases and liver diseases has been acknowledged.
Get detailed information on our products by downloading our catalog.
View Catalog
What are organoids?
Organoids are evolved spheroids that have been given functions as specific organs. It's not possible to culture actual organs because it is difficult to supply them with continuous nutrition. Organoids are smaller and simpler, so they are able to be cultured. The difference between organoids and spheroids is that while spheroids are lumps of 3D-cultured cells, organoids are more complex tissues composed of multiple cells that function as an organ.
Organoids are being researched for various organs, such as the brain, spinal cord, lungs, kidneys, stomach, and digestive tract. Even in the field of cancer research, organoids can be formed from the tumor cells of a patient and then used as the target model.
We’re here to provide you with more details.
Reach out today!

Spheroid culturing
The common culturing method for spheroids is monolayer culturing, in which cells are grown on a flat monolayer. As the proliferated cells cover the entire culture area, growth is inhibited or the cells die from contact inhibition. In vivo environment production is also difficult due to the low connection ratio between cells caused by planar growth. However, cells cultured through 3D proliferation maintain their shape in vivo. The high connection ratio between cells enables communication, and experiments similar to in vivo environments are possible. In consideration of the above, fully evaluating the effectiveness of medicines is sometimes not possible with single-layer cultured cells, which can be problematic.
Generally speaking, spheroids generated in 3D cultures should be uniform in number of constituent cells, size, and shape. Being able to generate large quantities of spheroids easily is also important.
This section introduces the three most commonly used culture methods currently in use: non-adhesive surface cell culturing, hanging drop cell culturing, and rotary cell culturing.
Non-adhesive surface cell culturing
Non-adhesive culture plates offer improved surface hydrophilicity for cell culturing. Cells dropped onto the non-adhesive plate float in the medium and adhere to each other to form a spheroid. The plate can also be shaken to increase the probability of contact between the cells. This method can be used to create large amounts of spheroids by increasing the size of the culture plate.
Hanging drop cell culturing
In this method, suspended cells are dropped onto the lid of the culture plate, and the droplets that rise due to surface tension are used for culturing. As the cells are pulled downward by gravity, they adhere to each other to form spheroids. This method makes it possible to create spheroids with a uniform number of cells.
Rotary cell culturing
This method involves rotating the culture chamber to create spheroids. By rotating the chamber, the suspended cells come into contact with each other evenly, enabling the creation of spheroids that are relatively uniform in both the number and size of cells. This method is becoming a popular choice for culturing large quantities of spheroids.
We’re here to provide you with more details.
Reach out today!

Example of spheroid observation
In the following example, KEYENCE's All-in-One Fluorescence Microscope BZ-X800 is used to observe a HEK293 spheroid that has been cleared through the use of Scale, a reagent that turns tissue transparent. Dead cells are colored red and fluoresce, making it possible to observe the distribution of dead cells, as well as the increase and decrease of them when the conditions are changed.
Conventionally, a laser confocal microscope was commonly used to observe spheroids and organoids, particularly given their 3D structure. However, there were several limitations with this approach, such as: 1) imaging took too long, 2) equipment was difficult to operate, and 3) cells were photobleached.
The optical sectioning function of the BZ-X Series can be used to observe thick samples without the influence of scattered or background fluorescence and do it much faster, easier, and with less damage. Compared to the standard wide-field image below, you can see just how clear of an image can be produced when using this function.
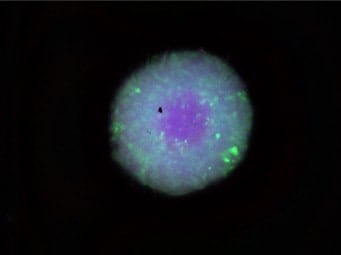
Normal observation
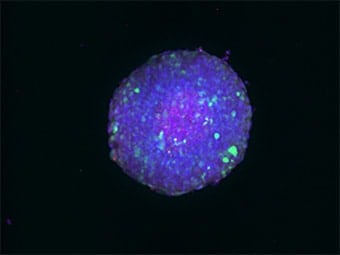
Sectioning observation
Using the All-in-One Fluorescence Microscope BZ-X800
- With an integrated darkroom chamber and fully-motorized operation, users can perform imaging tasks much more quickly.
- KEYENCE's proprietary optical sectioning method captures clear, fully-focused images even on thick tissues.
- The microscope has been developed with the utmost concern for cell damage, making it possible to observe the entire specimen with minimal photobleaching.
- A fully-focused, high-resolution 3D image can be created to clearly observe localization.


