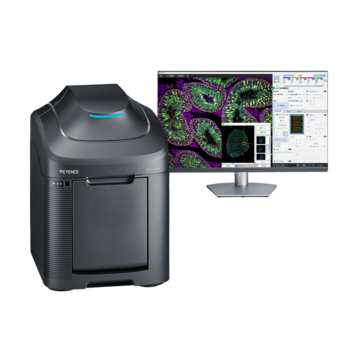
Fluorescence Microscopes
This page explains the place of emulsions among colloidal dispersion systems, as well as types of emulsions and their characteristics, double emulsions and their advantages, and how to assess them.
Get detailed information on our products by downloading our catalog.
View Catalog
What are emulsions?
Definition of emulsion
An emulsion is a colloidal dispersion system where larger droplets of the dispersed material (dispersed or discontinuous phase) are dispersed in the dispersion medium. Dispersed liquid materials are dispersed in a liquid dispersion medium without dissolving into each other. The process of forming an emulsion from two liquids is called emulsification. In general, a surfactant (emulsifier), which has both hydrophilic and lipophilic properties, is used to stabilize unstable emulsified state.
Difference between emulsions and suspensions
A suspension is another kind of dispersion system that has a liquid dispersion medium. The greatest difference between suspensions and emulsions is that the dispersed material is liquid in emulsions and solid in suspensions. Examples of suspensions include muddy water, ink, general paint, and toothpaste. In food, soups and sauces are generally suspensions. When gas is dispersed in a liquid dispersion medium, it is called a foam, some common examples of which are soda, meringue, and shaving foam.
Surfactant micelles in emulsions and their properties
An aggregate of surfactant molecules dispersed in a liquid forming into the size of a colloid is called an association colloid (micelle). Soap is a typical example of a substance that has a micelle structure. Surfactant molecules in detergent form clusters when they reach a certain concentration, becoming rod or plate-like micelles.
Types of micelle structures found in surfactants include (ordinary) micelles, where the hydrophobic tails assemble into a core in water with the hydrophilic side facing out, and reverse micelles, where the clusters have a hydrophilic core and the hydrophobic side facing out. These micelles give emulsions their emulsifying, detergency, solubility, and amphipathic molecular properties. For example, the oil-removing performance of detergent or face wash is delivered by the hydrophobic groups surrounding the oil in the core at a certain concentration and forming a micelle (emulsification). On the other hand, skin lotions can moisturize skin by the hydrophilic groups in a reverse micelle surrounding the moisture and emulsifying.
We’re here to provide you with more details.
Reach out today!

Types of emulsions
Emulsions, which are a mixture of two liquids that do not dissolve into each other, such as water and oil, are classified by which material is the dispersed material and which is the dispersion medium.
In the case of water and oil, the emulsion takes one of two compositions: Oil-in-Water (O/W), in which water is dispersed in oil, or Water-in-Oil (W/O), in which oil is dispersed in water. Examples of W/O emulsions include dairy products like butter and margarine, and creams in cosmetics. Mayonnaise and fresh cream, and skin cream in cosmetics are O/W emulsions.
Turning a W/O emulsion into an O/W emulsion and vice versa by means of agitation, temperature change, or use of surfactants is called phase inversion. For example, phase inversion occurs when making butter from cream. Other compositions, such as O/O and W/W emulsions, are also possible by stabilizing the emulsion with solid particles that adsorb onto the interface between the two phases (Pickering emulsions). In some cases, a co-surfactant is used to improve the stability and/or viscosity of the emulsion.
In recent years, synthetic techniques have been used to create double emulsions (also called emulsions of emulsions or multiple emulsions), such as W/O/W and O/W/O emulsions, which will be explained later. Microemulsions, which have fine emulsion droplets of the same size as those in the inner water phase of a double emulsion, are also garnering attention. The technologies used to compose and control a variety of emulsions made of non-combining substances are applied in many areas, including cosmetics, food, and pharmaceuticals. In the cosmetics industry, emulsion technologies are used in moisturizing creams and serums for the skin to control skin affinity, appearance, and texture. For food, studies are underway to apply such technologies to controlling flavor and calories. There is active research in the medical industry aimed at the use of emulsions in drug delivery systems (DDS), which precisely control the movement of drugs within the body so as to deliver them to the target location through the use of emulsions as drug carriers.
Examples of application and advantages of double emulsions
The larger the emulsion droplets of the material dispersed in an emulsion, the more strongly that substance’s characteristics will be expressed. For example, in food, flavor can be controlled by adjusting the size of the emulsion droplets. In general, larger oil droplets in an O/W emulsion tends to increase costs. Amid the increasing demand for low-calorie foods and beverages, achieving the intended flavor may also result in increased use of oil, thus making the food higher in calories. Double emulsion can be used as a method to resolve issues such as these.
For example, encapsulating water inside oil droplets can increase the apparent size of the oil droplets. Compositions like this are called Water-in-Oil-in-Water emulsions (W/O/W emulsions) due to the encapsulation of water inside the oil droplets in an O/W emulsion. This makes it possible to reduce the total amount of lipids used, helping to reduce both costs and calories without having to sacrifice taste. On the other hand, Oil-in-Water-in-Oil emulsions (O/W/O emulsions) can contain more moisture, which can be used in skin care cosmetic products to achieve a non-greasy, refreshing texture.
Despite the benefits, it is difficult to adjust the production conditions required for double emulsion products. Heat treatment can cause instability and destroy the double emulsion composition. Advanced research and development, sophisticated production techniques, and stable production conditions must be established to maintain a stable emulsion state not only during production, but also up to the point of consumption or use by consumers.
Emulsion (O/W)
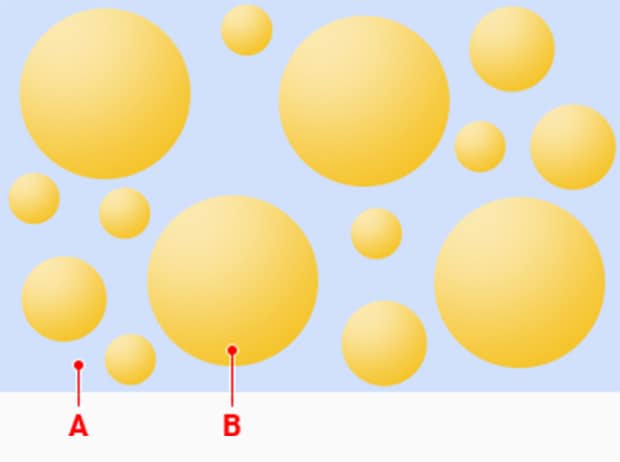
A: Water phase B: Oil phase
Double emulsion (W/O/W)
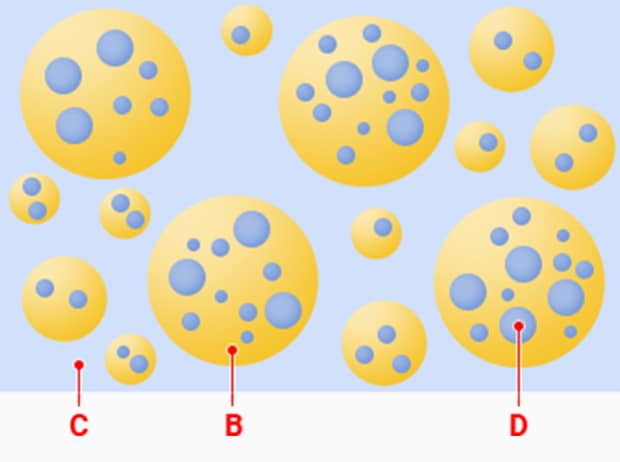
B: Oil phase C: Outer water phase D: Inner water phase
We’re here to provide you with more details.
Reach out today!

Stability assessment of emulsions
Emulsions are used for products in many areas, including cosmetics, food, and pharmaceuticals. These products must maintain their dispersion state during the period in which they are being used by consumers, as well as during production and after shipment. Long-term stability is required in particular with products like cosmetics and condiments, which are used over a certain period of time. This section explains the issues in stabilizing emulsions, effective solutions, and how to assess emulsion stability.
Issues in stabilizing emulsions
Dispersion systems such as emulsions are generally in a state of non-equilibrium. In most cases they are unstable, with a high interface energy between the non-combining liquids, causing the components to separate over time—this is a problem for maintaining the quality of emulsion products. Breaking an emulsion into its component phases is called demulsification. Demulsification may occur when water droplets settle down in an emulsion (sedimentation) or when oil droplets rise in the water phase (creaming), both of which are driven by the density difference between the inner and outer phases of the emulsion. When sedimentation or creaming occurs, if the intensity of the interfacial film is too low, the interface breaks and the inner phase droplets combine to form greater droplets, leading to a separation of the emulsion into water and oil phases.
Stabilizing emulsions
In order to maintain the stability of high-energy emulsions over an extended period of time, measures must be taken to prevent demulsification. For example, identifying the speed of sedimentation or creaming that occurs from the density difference between the inner and outer phases allows for demulsification to be controlled by resolving this density difference by adjusting the viscosity of the dispersion medium or reducing the droplet size of the dispersed material. Other countermeasures include adding electrolytes (ionic surfactants) to decrease electrostatic repulsion, which in turn prevents dispersed droplets from coalescing due to ionic absorption. In the case of O/W emulsions, a co-surfactant can be effectively used to prevent the breaking of the droplets’ interfacial film, thus suppressing the tendency to combine into each other. There are different types and compositions of emulsions, so measures suited to each particular product should be taken to maintain quality and ensure the stability of the emulsion.
Assessing the stability of emulsions
The stability of an emulsion can be assessed by observing its appearance. For example, observation of the droplet size of the dispersed material and the clarity of the emulsion are some of the key criteria for determining the stability.
Long-term stability of emulsification and dispersion is required in most emulsion products, including food, pharmaceuticals, and cosmetics. For example, spending a year to assess the one-year stability of an emulsion will require too much time spent on R&D and quality control—so it is necessary to take measures such as intentionally applying demulsifying stress to the product to shorten the time required for stability assessment. An example would be to use centrifugal separation to examine aggregation intensity and shaking to check for re-dispersion by measuring the turbidity in order to assess the stability of liquid foods or beverages containing milk constituents. Another approach is to assess the stability of dispersion by measuring the turbidity of the bottom layer of the dispersion system after leaving it for a certain period of time. Observing the appearance to examine the dispersion state after a certain period has elapsed and additionally applying stress in order to perform measurements and analysis allows for more detailed assessment. The next section explains how to observe and analyze emulsions, including double emulsions, for stability assessment, along with the problems that may be encountered and how to work around them.
We’re here to provide you with more details.
Reach out today!

Observing emulsions
For research and development of the stability of emulsification and dispersion, and to derive stable production conditions or to accurately determine the status when various treatments have been applied, a microscope must be used to directly observe the status of emulsions. High resolution is required for accurate imaging of the structure, and particularly double emulsions, which have minuscule droplets (inner water phase), are not observable with standard optical microscopes due to resolution limitations. Double emulsions consisting of water and oil phases are also difficult to observe using a scanning electron microscope (SEM) due to the need for vacuum.
Microemulsions, a recently established composition, have minuscule droplets of dispersed material with a diameter anywhere from the submicron order up to several micrometers, which is about the same size as those in the inner water phase of a W/O/W double emulsion. While a microemulsion has its dispersed material spread across a wider area, a double emulsion has the inner water phase existing in a relative aggregated state inside oil droplets, which makes the latter far more difficult to observe despite the similar sizes of the observation targets.
Emulsions in which spherical structures are dispersed over the entire field of view are susceptible to the impact of aberration. Conventional lenses with significant aberration cause color shifting and/or contour distortion in the outer perimeter of the field of view, preventing accurate profile observation and size measurements.
Observing emulsions (double emulsions) using a fluorescence microscope
KEYENCE’s BZ-X Series All-in-One Fluorescence Microscope uses Plan Apochromat oil-immersion lenses, which have the highest numerical aperture (NAs) and least aberration among all optical lenses for optical microscopes. This allows for high-resolution observation of objects within a 100 μm range in the Z direction of the cover glass to the back side of the cover glass. This means that the BZ-X Series can offer accurate, high-definition observation of emulsion dispersion as well as the contour and profile of the fine inner phase of double emulsions.
The BZ-X Series also supports observation at high contrasts for stained microscopy. For example, staining the oil phase of the emulsion with Oil Red O and the water phase with food coloring allows for distinction of the phases at a high contrast. You can also stain just the oil phase to clearly observe the region other than the inner water phase of the droplets with red fluorescence.
The BZ-X Series can easily achieve high-precision measurement and analysis of the minuscule inner phase droplet size of double emulsions, not to mention droplet count, longest and shortest diameters, and area in the field of view. This contributes to quick, quantitative assessment of the stability of emulsification and dispersion in quality assurance and control during production as well as during research and development. Additionally, the BZ-X Series offers a variety of observation approaches and quantitative analyses in a single device, helping save space by reducing the total equipment footprint.
W/O/W emulsion (100x)
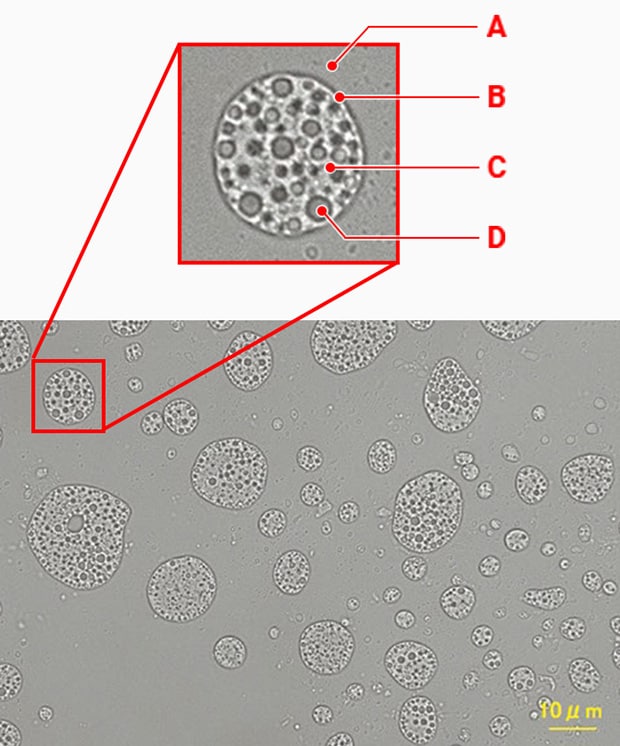
A: Outer water phase B: Oil droplet C: Oil phase D: Inner water phase
O/W emulsion (100x)
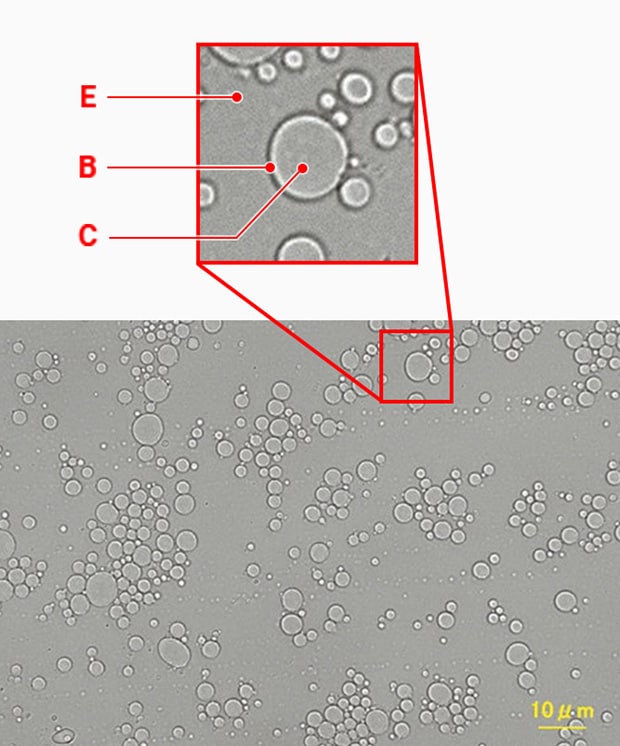
B: Oil droplet C: Oil phase E: Water phase
Using the All-in-One Fluorescence Microscope BZ-X Series
- Using the highest-resolution oil immersion lens (100x) with the lowest aberration among all objective lenses for optical microscopes, the BZ-X Series offers high-resolution observation of even minuscule contours and accurate profiles of the inner water phase of double emulsions.
- The BZ-X Series can achieve easy, accurate measurements of droplet counts, longest and shortest diameters, area in the field of view, and even the size of miniscule inner water phase droplets in double emulsions for quantitative assessment.
- Even with stained oil/water phase in emulsions, the BZ-X Series offers clear, high-contrast images for observation.
- The BZ-X Series offers a variety of observation approaches and quantitative analyses in a single device, so it helps save space.