Industrial Laser Marking Systems / Laser Markers
Types of Lasers: 8 Common Classifications of Laser
-
Tags:
- Automotive , Medical , Semiconductor
Key Takeaways
- Different types of lasers are classified by their gain medium: solid-state, gas, and liquid systems.
- Fiber lasers offer minimal maintenance and high reliability for metal processing applications.
- CO2 lasers excel at marking and cutting organic materials with their longer wavelength.
- UV lasers provide cold marking capabilities for heat-sensitive materials.
- Wavelength, power output, and material compatibility determine the best laser choice.

The most popular types of lasers used in industrial settings are described in this article. You'll discover the differences between solid-state and gas lasers, which wavelengths are ideal for particular materials, and how to adapt laser technology to your production needs.
Dating back to 1960, when Theodore Maiman invented the first laser, this form of light has evolved into a tool used across industries. From medical treatment to manufacturing processing, the power of lasers is used across a myriad of industries and for many types of applications.
When most think of lasers, the ubiquitous red light lasers often come to mind. However, there are many different types of lasers. In fact, there are over eight types of lasers across three categories! In this blog, we'll explore each category of laser, what the category entails, uses for lasers, and eight different types of laser machines that make up these categories.
How Industrial Lasers Are Classified
Based on their gain medium, industrial lasers can be divided into three primary groups: liquid, gas, and solid-state systems. Every category has special benefits for particular production procedures. Depending on the medium, laser marking methods range greatly among these categories in terms of beam quality and power output.
The choice between types of laser machines depends on material properties, precision requirements, and production volume. Manufacturers must consider wavelength compatibility with their target materials when selecting equipment.
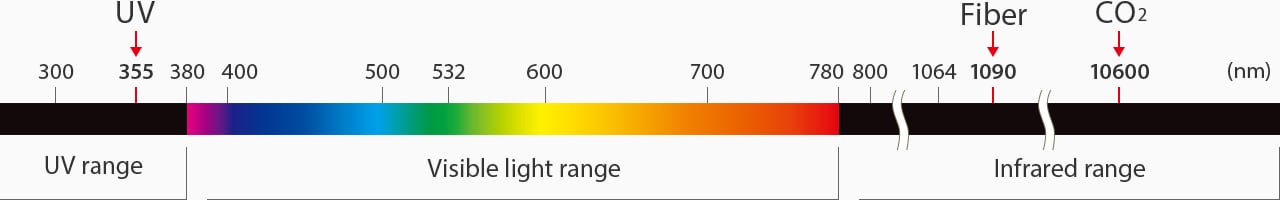
Light wavelength distribution map
Solid-State Lasers
Solid-state lasers are types of lasers that use a solid material as its gain medium. The most common solid-state laser media are yttrium aluminum garnet (YAG) and yttrium vanadate (YVO4). When a laser diode shines light on the doped material, the neodymium atoms are excited and emit light. This light is then amplified by the material, focused and emitted as a laser beam.
YVO4 requires large amounts of energy to produce a stable high-output energy amount. This medium has recently taken over in popularity compared to YAG mediums. It's known for assisting in the high-quality marking of metals, plastic, and ceramic.
YAG mediums require less energy than YVO4 lasers, which is why they reigned popular as the universal marking structure for so long. However, because of their need for less energy, this structure is not as stable as YVO4. YAG is known for more brute force and faster marking.
Fiber lasers dominate modern production environments due to their exceptional reliability and minimal maintenance needs. These systems deliver consistent beam quality over thousands of operating hours. Their compact form allows easy integration into existing production lines without major facility modifications.
Within the solid-state laser category are three different types of lasers: Green, UV, and Fiber.
Green Laser
Green lasers, also known as second-harmonic lasers, are notable for their specialized ability to process at a micron level. The laser begins at a 1064 nm wavelength and then passes through a nonlinear crystal for conversion into a 532 nm wavelength. With the much shorter wavelength that appears green to the human eye, intricacy is a green laser's special power. Green lasers are used as pointing devices and for micron-level marking and processing.
UV Laser
UV lasers, also known as third-harmonic lasers, are a specialized type of solid-state laser that uses “cold marking.” Cold marking refers to a UV laser's strength of absorption that produces very little heat when marking. These lasers start at a 1064 nm wavelength and then pass through two nonlinear crystals to transition to a 355 nm wavelength.
Because of the extremely small beam, small processing and minimal damage are two key components of a UV laser. UV lasers are powerful enough to create high contrast marks on highly reflective materials like copper, gold, and silver.
Fiber Laser Marking: High-Speed Metal Processing
Solid-state fiber lasers are highly regarded for their high average power, fast speed, and excellent cooling efficiency. With the ability to mark on metal, plastic, and ceramic with a 1030-2100 nm wavelength, it's even more impressive that fiber lasers can engrave, anneal, mark, etch, cut, and remove burrs. Fiber lasers have the smallest marking unit size, making them the easiest to retrofit or integrate in production settings with limited space.
Fiber systems handle demanding production schedules without frequent downtime. The sealed optical path prevents contamination that plagues open-beam configurations. Operators spend less time on calibration and more time running parts. Metal marking applications benefit from fiber technology's ability to create permanent, high-contrast identification codes that withstand harsh environments.
Fiber lasers have an edge over earlier technologies due to their energy efficiency. Wall-plug efficiency is 30%, while CO2 systems have an efficiency of 10%. Over the course of the equipment's lifetime, lower power consumption results in lower operational expenses. Facilities also appreciate the reduced cooling requirements, which simplify installation planning.
Gas Lasers: Versatile Industrial Applications
Gas lasers are used for machining and marking applications like marking labels, etching plastic and resins, processing, and cutting. These laser types use gas as the medium instead of a solid or liquid. The four types of gas lasers are CO2, He-Ne, excimer, and argon.
Gas systems produce several sorts of laser beams based on the gas composition employed. Each arrangement generates various wavelengths that are best suited to specific industrial applications. Gas purity and pressure must be monitored to ensure consistent output quality. Replacement intervals vary depending on system type and usage intensity.
CO2 Laser Marking: Ideal for Organic Materials
CO2 gas lasers have the longest wavelength compared to fiber, YAG, and YVO4. CO2 lasers have a wavelength of 10600 nm. Because of the long wavelength, there is more heat transfer than other lasers. Moreover, CO2 lasers are commonly used for cutting materials.
CO2 lasers excel at cutting plastic materials and processing organic compounds. Their longer wavelength generates more heat, which penetrates materials differently than shorter wavelengths. These systems require proper ventilation and gas supply management for optimal performance.
Packaging industries rely on CO2 technology for laser marking dates, lot codes, and graphics on cardboard, wood, and paper products. The infrared beam creates clean marks without contact, preventing material distortion. Food and beverage manufacturers use these systems for expiration date coding on bottles and containers.
Maintenance requirements include periodic gas refills and mirror alignment checks. Optics cleaning schedules depend on environmental conditions in the production area. Dust and airborne particles can degrade beam quality if not properly managed.
He-Ne Laser: Stable Beam for Precision Alignment
He-Ne gas lasers have a wavelength of 630 nm and appear red to the human eye. He-Ne lasers are commonly known for their use as pointing devices in classrooms or during presentations. These laser types are also used in store barcode scanners and measurement devices. Unlike many other types of lasers, He-Ne lasers do not do manufacturing processing.
He-Ne systems offer affordability for basic applications but lack the power needed for heavy manufacturing. Their visible red beam makes alignment and setup straightforward for operators.
Surveying and construction teams use He-Ne lasers for establishing level reference lines across job sites. The stable, continuous beam maintains accuracy over long distances. Machine tool manufacturers incorporate these lasers into equipment for precision positioning tasks.
Excimer Laser: Ultra-Fine Micro Processing
Excimer gas lasers use inert gas and hydrogen gas to produce a short UV wavelength of 193 nm. These lasers are commonly used for dermatology and optometry treatments. In fact, excimer lasers are proven to treat skin conditions like alopecia, psoriasis, leukoderma, and more. These lasers are also used in optometry to vaporize the lens of human eyes.
Excimer technology is essential to photolithography procedures in semiconductor fabrication. The short wavelength makes it possible to transfer patterns at the nanoscale sizes needed for contemporary microchips. This level of precision in laser etching propels advancements in processing power and device downsizing.
Argon Laser: High-Energy Visible Light Applications
Similar to excimer lasers, argon gas lasers are also used for various health conditions, scientific applications, and biomedical research. Argon lasers use a 488 to 514 nm wavelength, and one popular type of argon laser treatment is for glaucoma or diabetic eye disease treatment.
Research laboratories employ argon lasers for fluorescence microscopy and flow cytometry. The blue-green output excites fluorescent markers in biological samples. Spectroscopy applications take advantage of the pure wavelength for chemical analysis work.
Liquid Lasers
Dye Laser
Dye lasers are liquid lasers with a wavelength of 330 to 1300 nm, commonly used for scientific applications. These lasers use organic liquid dyes to power their wavelengths and produce fluorescent light.
Tunable wavelength capability sets dye lasers apart from fixed-wavelength systems. Researchers adjust output across a broad spectrum by changing dye solutions. This flexibility supports diverse experimental needs without purchasing multiple laser types. Academic institutions value this adaptability for teaching and investigation purposes.
UV Lasers: When and Why to Use Them
When it comes to fragile materials that are unable to withstand heat damage, UV lasers offer unrivaled precision. When marking, its high absorption capabilities keep materials from warping or discoloring. UV systems are used by electronics makers to identify components without sacrificing circuit integrity.
These systems work best on plastics, glass, and sensitive substrates where thermal effects must be minimized. Types of lasers in the UV category require more careful handling due to their invisible beam properties.
Medical device manufacturers choose UV technology for plastic marking on implantable components. Biocompatibility requirements prohibit marks that introduce contaminants or alter surface chemistry. Cold ablation removes material at the molecular level without creating heat-affected zones.
Printed circuit board fabrication uses UV lasers to drill micro-vias connecting internal layers. Hole diameters measure just micrometers across. Traditional mechanical drilling cannot achieve this scale without damaging delicate copper traces.
Choosing the Right Laser for Your Application
When choosing an industrial laser, compatibility of materials, manufacturing speed requirements, and quality standards must all be carefully considered. Every kind of laser has unique benefits for particular uses. Automotive manufacturers often prefer fiber systems for their durability and speed.
UV systems are commonly used by medical equipment manufacturers for biocompatible branding that poses no risk of contamination. The choice has a major effect on both the initial outlay and ongoing operating expenses.
Production volume projections influence equipment selection more than purchase price alone. High-throughput facilities justify premium systems through reduced per-part costs. Lower volume operations may find benchtop models adequate for their needs. Trial runs with sample parts reveal whether a laser meets quality expectations before committing to purchase.
Ceramic marking presents unique challenges due to material hardness and thermal sensitivity. Different types of lasers produce varying results on alumina, zirconia, and other ceramic substrates. Test marking helps identify optimal parameters for each material grade.
Wavelength, Power & Budget Factors
Wavelength determines material absorption characteristics and achievable mark quality. Shorter wavelengths generally provide finer detail but may require higher initial investment. Power requirements scale with production volume and material thickness.
Budget considerations must include consumables, maintenance schedules, and energy consumption over the equipment’s lifetime. Laser marking technologies continue advancing, making older systems less cost-effective over time.
Labor savings from automation should be taken into consideration when calculating return on investment. By reducing the requirement for staff, a laser labeling system that does away with manual application stages pays for itself. By averting recalls and warranty claims, traceability enhancements also lower expenses.
Upgrade paths matter for growing businesses. Expanding capacity is possible with modular systems without having to replace entire installations. Updates to software can give current hardware new features and increase its useful life.
3-Axis CO2 Laser Marker ML-Z9600

World’s first 3-Axis control laser marker
3-Axis Fiber Laser Marker MD-F3000

World’s smallest fiber laser marking unit
Telecentric Laser Marker MD-T1000

World’s first green laser marker with a telecentric lens
3-Axis UV Laser Marker MD-U2000

World’s first UV laser marker with 3-Axis control
3-Axis Hybrid Laser Marker MD-X2000/X2500

World's first laser marker with full-field auto-focus
Contact us to learn more about how our advanced technology can help take your business to the next level.
Contact Us
Frequently Asked Questions
How Does Laser Wavelength Affect the Quality of Marking?
Shorter wavelengths (e.g., UV) produce high-resolution marks on delicate materials. Longer wavelengths like CO2 provide deeper penetration for cutting applications.
Are Certain Lasers Better Suited for Automated Production?
Solid-state lasers are often preferred due to low maintenance and integration flexibility. Their design lacks the same maintenance requirements found in CO2 systems.
Can One Type of Laser Handle Both Marking and Cutting?
It is possible, but it depends on the application requirements. For example, a UV laser can result in high quality marking, but a CO2 laser's higher power may be preferred for faster cutting time.
What Industries Benefit Most From Fiber Lasers?
Automotive, aerospace, and electronics industries rely heavily on fiber lasers. Their precision and reliability meet the strict quality standards required in these sectors.
What Are the Main Types of Laser Beams?
The three main types of lasers are solid-state, gas, and liquid lasers, each using different gain mediums to produce laser light. Solid-state includes fiber and UV types, while gas encompasses CO2 and argon systems.
How Do Solid-State Lasers Differ From Gas Lasers?
Solid-state lasers use crystalline or glass materials as their gain medium, while gas lasers rely on gas mixtures. Solid-state systems typically require less maintenance and offer more compact designs.
We’re here to provide you with more details.
Reach out today!







