Fluorescence Microscopes
Analysis with calcium imaging
In recent years, the number of researchers performing calcium imaging (Ca imaging) using a fluorescence microscope has increased.
Calcium imaging is a method in which the flow of intracellular calcium is measured so as to directly observe the calcium signaling of active neurons. This method is widely used in applications such as the research of brain cells.
It is common knowledge that calcium is an extremely important component for vertebrates that live on land. Calcium also serves an important role in the formation of strong teeth that enable the ingestion of a variety of plants and animals. Furthermore, calcium takes part in many phenomena of life within organisms including the secretion of hormones and neurotransmitters, the contraction of muscles, and the moldable changes of neurons. By observing the calcium ion concentration of neurons that are active within organisms, calcium imaging enables detailed measurement of cell activities in which calcium ions take part.
Get detailed information on our products by downloading our catalog.
View Catalog
Calcium imaging principles
When carrying out calcium imaging, use a fluorescence microscope and a fluorescence plate reader to perform observation. Introduce protein, whose fluorescence intensity changes upon bonding with calcium ions, and a calcium fluorescence indicator into the cell, and then use the changes in fluorescence intensity to detect the changes in calcium ion concentration.
The calcium fluorescence indicator is extremely weak, typically requiring a powerful laser to excite the indicator in order to observe it. This high-intensity laser accelerates the death of the cell and makes achieving successful calcium imaging difficult.
We’re here to provide you with more details.
Reach out today!

Common failures of calcium imaging and corresponding countermeasures
Common failures of calcium imaging include the cell weakening, the image going out of focus, and the cell moving out of the field of view. These failures are also common when performing live cell imaging using a fluorescence microscope.
For example, if you start calcium imaging and wait for some time before checking the cell status, the cell may have weakened or died.
One major cause is cell damage caused by the powerful excitation light (phototoxicity).
Some countermeasures against this problem include:
- Using a camera with as high a sensitivity as possible.
- Using gain and binning.
- Configuring the settings such that even weak fluorescence can be detected.
- Reducing the intensity of the excitation light as much as possible.
- Turning off the excitation light when not capturing images.
Additionally, the phenomenon of the image going out of focus may occur during calcium imaging. Possible causes of this include: the cell moving due to temperature drift, the settings being incorrect, and the cell moving (especially vertically) during cell division. Most researchers who perform imaging with a fluorescence microscope have experienced these issues.
As a countermeasure, the auto focus function or the Z-stack function can be used to prevent the image from going out of focus.
We’re here to provide you with more details.
Reach out today!

Calcium imaging observation example
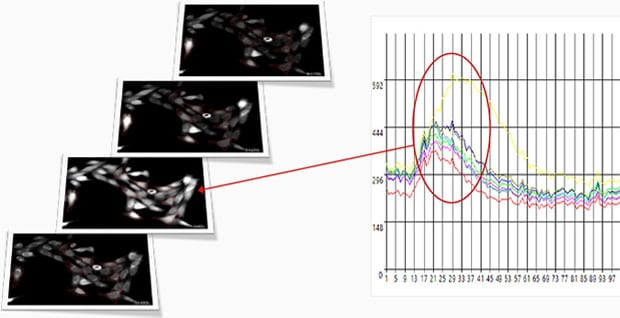
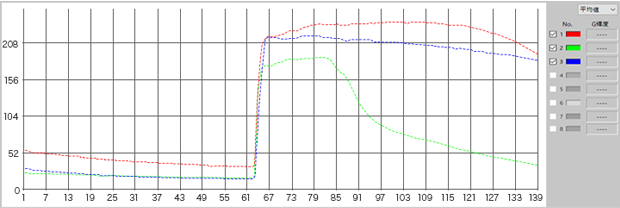
Fluorescence intensity change via the addition of a drug
Calcium imaging of a myoblast cell (CPA, Fluor8, 5 second intervals, 10 minutes)
G CaMP calcium imaging test



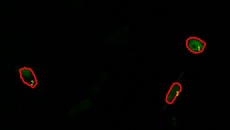
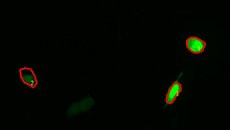
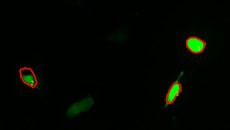
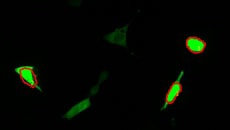
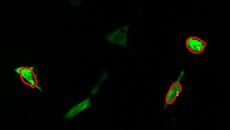
Using the All-in-One Fluorescence Microscope BZ-X
- No darkroom required and easy operation for extremely clear microscopy.
- A highly sensitive optical system composed of a cooled camera and a dedicated lens makes it possible to obtain high-resolution fluorescence images with minimal cell damage.
- Multiple samples can be time-lapse imaged at the same time, making it easy to perform comparative verification under the same conditions.
We’re here to provide you with more details.
Reach out today!

What is G CaMP?
This is one type of calcium sensor that is made of protein and is formed by bonding together a green fluorescent protein, a calmodulin, and a myosin light chain fragment via genetic engineering. G CaMP can be inserted into genes, thereby making it possible to perform calcium imaging on only the targeted structure or carcinoma through combination with a promoter that is specific to the structure or cell of the model animal.