Continuous Inkjet Printers / Case Coder
Materials and Products
Production Machines
Printing Applications
CIJ Coding Solutions for Pharmaceutical & Medical Device Packaging
Pharmaceutical packaging codes link each unit to manufacturing data, tracking the product through distribution, verification, and eventual use. These codes connect each unit to manufacturing records, distribution information, and downstream verification systems. In this context, pharmaceutical packaging inkjet coding becomes an integral part of the production process, not just a finishing step.
Continuous inkjet (CIJ) systems like the MK-G Series are commonly used as they support variable data printing at line speed while adapting to packaging formats that change frequently across product families. In addition, the CC-1000 Series excels in secondary packaging, offering a versatile solution for marking cartons and other outer packaging with high-quality, legible codes that maintain accuracy even at high speeds.

Barcode / 2D code
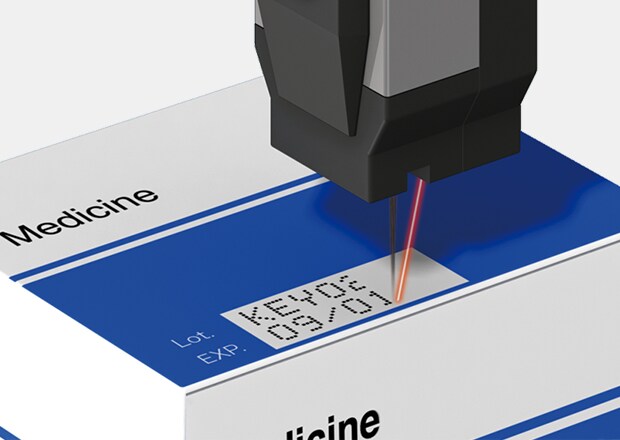
Installation example
We’re here to provide you with more details.
Reach out today!

Date Coding and Marking on Pharmaceutical and Medical Device Packaging
CIJ printers enhance productivity, tracking, and traceability on various packaging materials. This includes:
- Packaging films, pouches, and labels
- Carboard boxes
- PET bottles
- Blister Packs
- Cartons
- Glass bottle and ampoules

Pouches

Cardboard boxed

PET bottles

Heat-sealed packages

Cartons

Glass containers
Curious about our pricing?
Click here to find out more.

Reliable Inkjet Marking for Medical Products
Medical packaging lines have to apply codes while products are already in motion, leaving little opportunity for correction once marking begins. With containers passing through filling, sealing, and inspection stages without stopping, there are tight limits on how codes are formed.
CIJ printers are used in these conditions because labelings are typically generated without contact, reducing disruption on moving surfaces. When it comes to low-absorbency materials, ink drying and adhesion determine whether codes remain intact beyond the point of application. When ink selection aligns with the substrate and cleaning process, CIJ supports medical product traceability printing across downstream handling and secondary packaging.
Key Challenges in Pharmaceutical and Medical Coding
Pharmaceutical packaging introduces constraints that shift from one product format to the next; pharmaceutical packaging inkjet coding is being applied to small glass containers, coated cartons, or flexible films. This often takes place within the same production area and without the ability to stop the line for each packaging change.
Space constraints around filling and sealing equipment limit mounting options and access for adjustments. At the same time, serialized data and variable fields increase the importance of accurate message control. These factors place pressure on coding systems to operate predictably without introducing additional complexity to validated processes.
- Achieving Consistency Across Packaging Types
High-performance, adaptable coding technology for various packaging types offers seamless integration, minimal maintenance, product protection, and clear lot/date codes. - Meeting Regulatory Compliance
Adaptable, high-contrast, damage-free printing produces clear and compliant labeling on pharmaceutical and medical device packaging. - Ensuring Product Safety
Ensure product safety by providing consistent, high-quality, and legible prints for both OTC purchases and patient prescriptions. - Coding and Marking in High-Speed Production
KEYENCE CIJs offer easy integration and the ability to mark clear lot/date codes on a wide range of materials and shapes in even the most demanding production lines.
Barcode and 2D Code Integration
Different serialized identifiers that are printed on pharmaceutical packaging are read at multiple points during the production process. Code performance depends on uniform dot formation and contrast that stays within verification tolerances at line speed. Medical product traceability printing has to present a fine resolution across different materials without disrupting packaging flow. CIJ supports this level of consistency while operating in line with high-throughput packaging equipment.
Discover more about this product.
Click here to book your demo.
CIJ for Vials, Blisters, and Boxes
Pharmaceutical packaging inkjet coding lines rarely stop to accommodate format changes. Or, marking conditions that shift as products move through filling, sealing, and aggregation.
- Primary container printers, like CIJ for vials & syringes, are marked in tight spaces while still in motion.
- Foil-based packs may appear clean at print, then shift once heat sealing and cutting introduce stress to the packaging material.
- Secondary packaging carries variable data for distribution. If errors are detected here, it is typically no longer isolated to a single unit.
CIJ adapts to these conditions through parameter and consumable changes rather than mechanical reconfiguration. This helps keep coding aligned as products move beyond the print zone.
Why Continuous Inkjet Fits Healthcare Packaging
Many pharmaceutical packaging lines operate in controlled areas like cleanrooms or in completely automated environments. To comply with regulatory codes, access has to be limited.
In these spaces, even small interventions can mean extra steps for cleaning or documentation. CIJ printers are often set up to run through extended production windows with minimal touchpoints, so coding continues as containers move through filling and sealing.
MK-G Series
The MK-G Series is a self-troubleshooting continuous inkjet printer featuring automation, such as hands-free cleaning, maintenance, and consistent performance. Its ability to self-diagnose significantly reduces the time needed to recover from errors without waiting for a technician. For a comprehensive packaging solution, the MK-G Series serves as the primary packaging printer, while the CC-1000 Series excels as the secondary printer, offering reliable coding for outer packaging such as cartons and cases.
Contact the team at KEYENCE to achieve compliant packaging codes today.
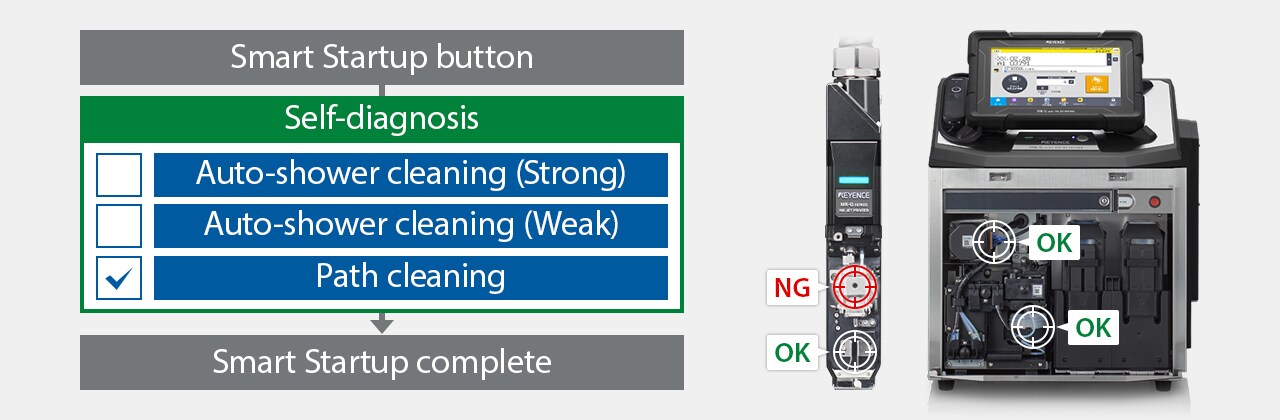
Using the MK Dock enables self-diagnosis and cleaning to ensure stable startup.
Contact us to learn more about how our advanced technology can help take your business to the next level.
Contact Us
FAQs About Coding, Serialization, and Marking on Packaging in the Pharmaceutical and Medical Device Industry
How Does the KEYENCE MK-G Series Marking and Coding Printer Specifically Address the Unique Needs of Coding on Pharmaceutical and Medical Device Packaging?
Promising a combination of benefits, MK-G Series printers can handle the wide range of coding requirements found in pharmaceutical and medical devices. Real-time quality checks, automated maintenance and diagnostics, and vast product compatibility handle many industry-specific needs. MK-G Series’ precise markings meet stringent formatting and content requirements set by regulatory bodies.
How Do Date Coding and Marking on Pharmaceutical and Medical Devices Contribute to Enhancing Anti-Counterfeiting Measures and Product Authentication?
This process ensures traceability through high-quality, reliable printed regulatory codes. Including expiration and manufacturing dates helps verify product authenticity, while maintaining high production throughput and efficiency.
What Are the Key Considerations When Implementing Marking and Coding Solutions onto Packaging Materials in the Pharmaceutical and Medical Devices Manufacturing Industries?
The inkjet coder should be able to seamlessly integrate into existing production lines. Throughput and marking speed are crucial to maintaining high production efficiency, while ease of maintenance and cleaning prevents downtime. Additionally, the solutions must comply with safety regulations to meet industry standards.
What Are the Main Challenges When Printing Date Codes on Pharmaceutical and Medical Device Packaging?
The main challenges include ensuring precise, durable codes that comply with regulations and serialization requirements. Maintaining high-speed production without compromising quality is also essential, and the ability to print consistently on various packaging materials adds to the complexity.
We’re here to provide you with more details.
Reach out today!

Related Downloads

This guide explains continuous inkjet (CIJ) printer applications together with pictures and illustrations. It contains many installation examples in various industries divided into food, medicine, and cosmetics; electrical machinery and electronics; and automotive, metal, and others. These examples show printing on targets specific to each industry.
Related Products
Applications
Materials and Products
- Glass Bottles
- Plastic Bottles/Containers
- Cans
- Wires, Cables, & Pipes
- Pouches
- Metal Parts
- Corrugate Boxes
- Packaging Film
- Kraft Packaging
- Folding Cartons
- Building Materials
- PCBs
- Cartons
- Pallets & Lumber
- Corrugate Trays
Production Machines
- Paper Bag Packaging
- Box Conveyor
- Automatic Palletizer
- Sealer/Taping Machine
- Bottle Filling Machine
- Flow Wrapper



![Process-specific Marking Applications [Food/Pharmaceutical Industry]](/img/asset/AS_133442_L.jpg)


